Did you know that 1 in 3 homes in Maine test too high for Radon?
Your exposure could equal smoking 2 packs of cigarettes a day!
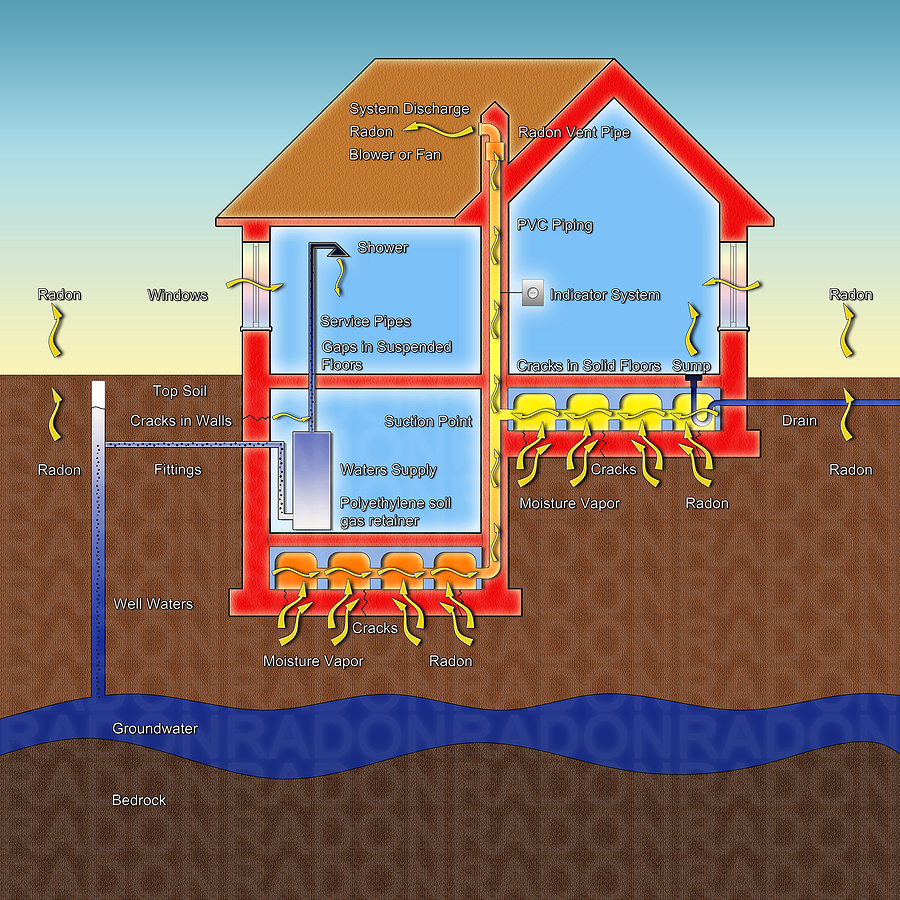
Radon is considered to be unsafe in any amount but the Environmental Protection Agency and the State of Maine consider a test level over 4 pCi/L (picocuries per liter) in the air and 4,000 pCi/L in water as an average level to be dangerous. If you are living in a home where you are constantly getting exposed to this level, you would have a higher radiation exposure than is permitted for nuclear power plant workers. More than 25% of the homes tested in Maine are above 4 pCi/L, and shockingly, it is not uncommon to see a house test over 50 pCi/L which, with annual exposure, equates to smoking two packs of cigarettes each day. Just because your neighbor’s house tests high, or low, does not mean your house’s will test the same so it’s very important to have your own home’s radon levels tested.
Millions of homes have radon levels that are too high, and are completely unaware of the danger.
Find out more about Radon testing on our Maine Radon Solutions website.